Audio By Carbonatix
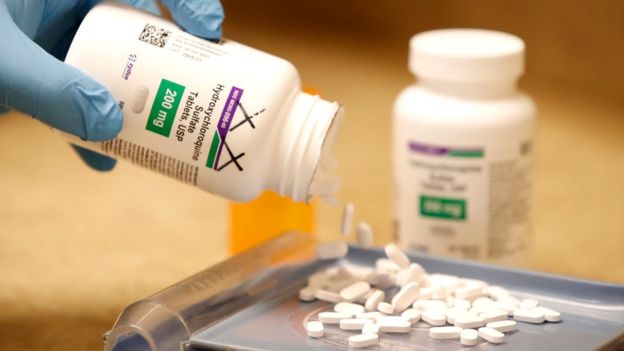
Testing of the malaria drug hydroxychloroquine as a possible treatment for coronavirus has been halted because of safety fears, the World Health Organization (WHO) says.
Trials in several countries are being "temporarily" suspended as a precaution, the agency said on Monday.
It comes after a recent medical study suggested the drug could increase the risk of patients dying from Covid-19.
President Donald Trump has said he is taking the drug to ward off the virus.
The US president has repeatedly promoted the anti-malarial drug, against medical advice and despite warnings from public health officials that it could cause heart problems.
Last week, a study in medical journal The Lancet said there were no benefits to treating coronavirus patients with hydroxychloroquine, and that taking it might even increase the number of deaths among those in hospital with the disease.
Hydroxychloroquine is safe for malaria, and conditions like lupus or arthritis, but no clinical trials have recommended its use for treating Covid-19.
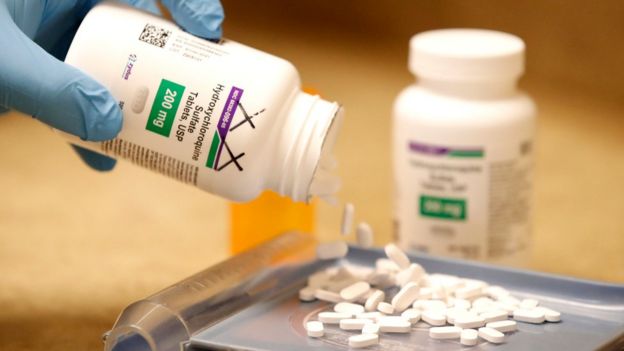
The WHO, which is running clinical trials of various drugs to assess which might be beneficial in treating the disease, has previously raised concerns over reports of individuals self-medicating and causing themselves serious harm.
On Monday, officials at the UN health agency said hydroxychloroquine would be removed from those trials pending a safety assessment.
The Lancet study involved 96,000 coronavirus patients, nearly 15,000 of whom were given hydroxychloroquine - or a related form chloroquine - either alone or with an antibiotic.
The study found that the patients were more likely to die in hospital and develop heart rhythm complications than other Covid patients in a comparison group.
The death rates of the treated groups were: hydroxychloroquine 18%; chloroquine 16.4%; control group 9%. Those treated with hydroxychloroquine or chloroquine in combination with antibiotics had an even higher death rate.
The researchers warned that hydroxychloroquine should not be used outside of clinical trials.
Latest Stories
-
I have supported highway authority financially to fix roads in my constituency – A Plus
21 minutes -
US, Iran fail to reach peace agreement after marathon talks in Pakistan
45 minutes -
ECG kicks off Phase Two of transformer upgrades at Lashibi; brief outages expected
1 hour -
Port crises loom as 11,000 drivers threaten four-day strike
2 hours -
A source of excellence across generations – Vice President Opoku-Agyemang lauds Mfantsipim
3 hours -
(Photos) Mfantsipim School launches historic 150th anniversary
3 hours -
Knights and Ladies of Marshall group backs Catholic Bishops’ stance on anti-LGBTQ+
4 hours -
Bright Simons writes: All the Filla in the Ibrahim Mahama/E&P – Gold Fields Saga
5 hours -
Monetise Idiocy In Ghana
5 hours -
The Ghanaian prophet and the mysterious death of his scottish wife Charmain Speirs
6 hours -
Nearly 400 sentenced in Nigeria for links to militant Islamists
6 hours -
Ghana’s recovery supported by gold strength despite global oil price pressures – Standard Bank Research
6 hours -
Methodist Church hails Mfantsipim@150; calls for “fresh consecration” to excellence
6 hours -
‘Excellence is our inheritance’ – Nana Sam Brew-Butler hails Mfantsipim’s 150-year reign in leadership
7 hours -
Kwaku Azar writes: A-G vs OSP
7 hours