Audio By Carbonatix
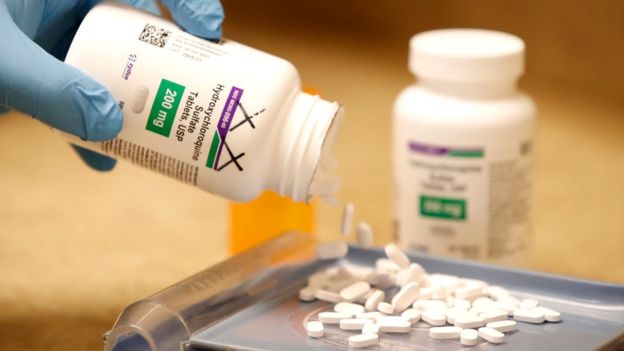
Testing of the malaria drug hydroxychloroquine as a possible treatment for coronavirus has been halted because of safety fears, the World Health Organization (WHO) says.
Trials in several countries are being "temporarily" suspended as a precaution, the agency said on Monday.
It comes after a recent medical study suggested the drug could increase the risk of patients dying from Covid-19.
President Donald Trump has said he is taking the drug to ward off the virus.
The US president has repeatedly promoted the anti-malarial drug, against medical advice and despite warnings from public health officials that it could cause heart problems.
Last week, a study in medical journal The Lancet said there were no benefits to treating coronavirus patients with hydroxychloroquine, and that taking it might even increase the number of deaths among those in hospital with the disease.
Hydroxychloroquine is safe for malaria, and conditions like lupus or arthritis, but no clinical trials have recommended its use for treating Covid-19.
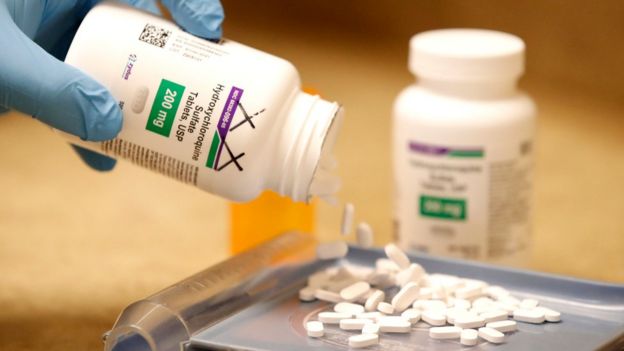
The WHO, which is running clinical trials of various drugs to assess which might be beneficial in treating the disease, has previously raised concerns over reports of individuals self-medicating and causing themselves serious harm.
On Monday, officials at the UN health agency said hydroxychloroquine would be removed from those trials pending a safety assessment.
The Lancet study involved 96,000 coronavirus patients, nearly 15,000 of whom were given hydroxychloroquine - or a related form chloroquine - either alone or with an antibiotic.
The study found that the patients were more likely to die in hospital and develop heart rhythm complications than other Covid patients in a comparison group.
The death rates of the treated groups were: hydroxychloroquine 18%; chloroquine 16.4%; control group 9%. Those treated with hydroxychloroquine or chloroquine in combination with antibiotics had an even higher death rate.
The researchers warned that hydroxychloroquine should not be used outside of clinical trials.
Latest Stories
-
Church of Pentecost supports over 2,000 BECE candidates in Obuasi with career guidance seminar
8 minutes -
Brandon Asante and Coventry all but promoted to Premier League despite Sheffield Wednesday draw
29 minutes -
GPL 2025/26: Late Kwartemaa strike downs Hearts in Tema
36 minutes -
Ghana Faces Sierra Leone Moment as Prosecutorial Powers come under strain
46 minutes -
Don’t consume fish or seafood from Tema Shipyard until further notice – FDA warns
50 minutes -
Why volunteering might be Africa’s most underrated career accelerator
57 minutes -
ActionAid Ghana raises concern over gender gaps in Feed Ghana Programme
59 minutes -
Windstorm wreaks havoc in Gushegu, displacing nearly 2,000 residents and damaging schools
1 hour -
Friends of Bridget Bonnie Marks her 35th birthday with donation to Kasseh Model Health Centre
2 hours -
From Ekumfi Kokodo to the Pulpit Stage: Essi Donkor’s gospel journey takes shape
2 hours -
Landfilling waste management creates no value, it’s an economic waste
2 hours -
Photos: Speaker Bagbin Commissions MPs constituency office under parliamentary decentralisation programme
3 hours -
Black Stars technical advisor Winfried Schäfer sacked as GFA shakes up backroom staff
3 hours -
Wenchi water project almost complete, critical to gov’t agenda – GWL MD
3 hours -
Anti-LGBTQ+ bill not part of government’s legislative agenda – Inusah Fuseini
3 hours