Audio By Carbonatix
Regulators in South Africa have recalled a batch of the popular Yaz Plus contraceptive pill, after a packaging mix-up which means the contraception could be potentially ineffective.
Manufacturer Bayer Ltd said women using pills from the affected batch should stop immediately and seek medical advice.
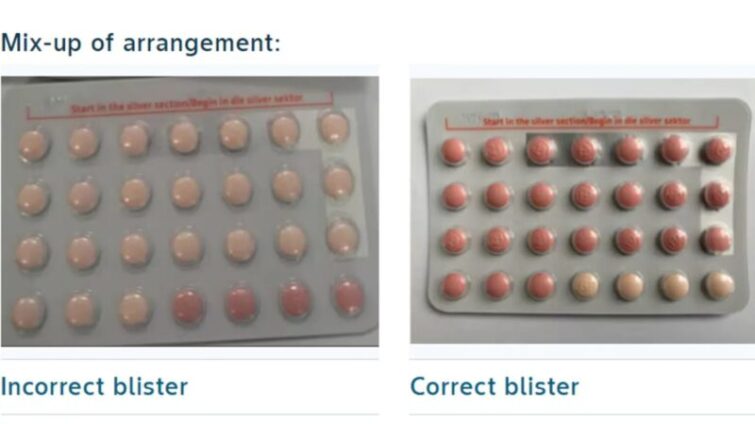
A packaging mix-up led to a number of blister packs carrying 24 inactive pills, instead of 24 hormone-containing active pills.
The issue affected only a limited number of packets in a specific batch labelled WEW96J, expiring in March 2026
The erroneous batch has been recalled by Bayer, in consultation with the South Africa Health Products Regulatory Agency, with the company stressing the "root cause" of the mix-up had been identified and dealt with.
A regular pack of Yaz Plus contraceptives contains 24 active pills containing hormones, which are pink in colour, followed by four hormone-free, inactive pills, which are light orange in colour.
In the recalled batch, a number of packs instead carried 24 hormone-free inactive pills and only four active hormone pills.
The concern is that a woman could be at risk of becoming pregnant having taken inactive pills believing she was taking effective hormonal contraception.
Bayer Ltd's recall notice says: "While only a limited number of packs from the respective batch is affected, as a precautionary measure, no tablets from these packs shall be used until you have consulted your healthcare practitioner, as they may potentially not provide the contraceptive protection you expect."
Anyone who has acquired a packet of pills belonging to the stipulated batch is advised to return the tablets to pharmacies for a replacement or refund.
Healthcare professionals, hospitals, pharmacies, doctors, nurses and wholesalers who have packets of the affected batch should also return them.
In a statement, Bayer Ltd said "The root cause for the mix-up of tablets in the packaging has been identified and corrective measures have been implemented".
The incident is limited to only one batch and no other batches are affected, the company said.
The company has set up a helpline for people with any further questions.
Latest Stories
-
What Is Wrong with Us: Why we keep uprooting young trees because they have not yet become forests
10 minutes -
Farmers are watching food rot – Group warns of deepening food glut crisis
1 hour -
Completed but locked: CDM slams gov’t over Weija Children’s Hospital
1 hour -
Pope Leo says AI must be ‘disarmed’ in first major teaching
4 hours -
Jordan leads star names at Guardiola leaving party
4 hours -
Allegri sacked after season of ‘unequivocal failure’
4 hours -
Man Utd captain Fernandes takes on Keane over ‘lie’
5 hours -
The rise of the fruit that tastes like custard
5 hours -
Ferrari unveils first fully electric car
5 hours -
Portable charger problems on flights ‘on the rise’, watchdog warns
5 hours -
Court rejects tendering of pictures in ongoing Boasiako illegal mining case
5 hours -
Indian billionaires buy foreign companies as growth slows at home
5 hours -
Mexico to host Iran for FIFA World Cup 2026
5 hours -
Absa Bank Ghana empowers businesses to navigate market risks
6 hours -
Moroccan Sahara: The preeminence and relevance of the autonomy plan highlighted in Verona
6 hours